 What is Ozone?
What is Ozone?
Ozone is often called nature’s purifier. It’s present at the seashore, in rainforests, and after thunderstorms. Most of us know it best as the protective ozone layer high in the atmosphere, but ozone—also known as activated oxygen or trivalent oxygen—is also created naturally through lightning, photosynthesis, or ultraviolet radiation from the sun.
Ozone forms when oxygen molecules (O2) are split into individual atoms, which then recombine with other oxygen molecules to create O3. The term “ozone” comes from the Greek word “ozein,” meaning “to smell.” In high concentrations, ozone has a faint bluish color, which is partly responsible for the blue appearance of the sky.
Discovered in the late 1800s, ozone is the second strongest oxidizer in the world. For more than a century, it has been used to purify water and eliminate odors in a wide variety of applications. Modern ozone generators mimic nature by creating ozone through high-voltage corona discharge—the same way lightning does during a storm.
How Does Ozone Eliminate Odors?
Ozone is unstable by nature, with a half-life of about 20 minutes. Once created, it quickly begins to break down, releasing a free oxygen atom. That atom either rejoins another oxygen molecule to form O2 or reacts with other gases in the air.
This powerful oxidizing reaction is what eliminates odors. For example, in a smoke-filled room, the odor comes from phenol gases clinging to the air and surfaces. When ozone is introduced, it attaches to those gas molecules, breaking them apart and destroying their odor-causing properties.
Benefits of Using Ozone
Ozone is a highly effective deodorizer because it doesn’t just mask odors—it destroys them at the source. It can eliminate:
- Tobacco smoke
- Pet and human odors
- Food-related smells
- Mold and mildew odors
- Garbage and renovation odors
- Many other unwanted scents
Unlike sprays or re-odorants, ozone neutralizes problem gases in the air, on fabrics, and on surfaces like walls and ceilings. Because it’s a free-floating molecule, it doesn’t require air to pass through a filter or machine to be effective, allowing it to reach areas other systems miss.
In addition, ozone generators also produce ions during corona discharge. These ions cause dust and other airborne particles to clump together and settle out of the air, leaving the environment fresher and cleaner.
What is a Hydroxyl Radical?
The hydroxyl radical (- OH) is the neutral form of the hydroxide ion (OH–). These diatomic molecules are extremely reactive and very short-lived, with a half-life of less than two seconds. First identified in 1963, hydroxyl radicals are often called the “detergent of the atmosphere” because they naturally help clean the air. By breaking down pollutants, they play a key role in reducing harmful compounds and even help eliminate certain greenhouse gases such as methane and ozone.
The Technology Behind Hydroxyl Generators
In the restoration and air purification industries, hydroxyl radicals are typically produced using UV light. UV light is divided into three major spectrums:
- UVA (315–400nm): Known as “black light,” safe for skin and eyes, and does not create ozone.
- UVB (280–315nm): The type of light used in tanning beds.
- UVC (100–280nm): Known as “germicidal” light. Direct exposure can be harmful to eyes and skin. Lamps in the 185nm range also generate ozone.
There are three main approaches to hydroxyl generation:
- Basic UVC Method: Some manufacturers use UVC lamps (185–254nm) without a catalyst and market them as hydroxyl generators. In reality, these are simply ultraviolet ozone generators and should be avoided.
- UVC with Catalytic Metals: Other systems use UVC in the 185–254nm range with catalytic metals such as titanium. These generate some hydroxyl radicals but also produce ozone, making them suitable only for unoccupied spaces.
- NASA-Developed Technology: The most advanced and trusted approach uses UVC light at 254nm to excite nano-particles of titanium dioxide, which generate hydroxyl radicals. This method is safe, effective, and produces no harmful ozone. It was developed with NASA and has been proven safe on the International Space Station. It is also FDA-certified as a Type 2 Medical Device, approved for hospital use—even in sensitive environments like neonatal and infant care units.
Why Hydroxyl Technology Matters
Hydroxyl radicals work continuously to neutralize odors, pollutants, and microbes in occupied spaces without the limitations of ozone. Their ability to safely and naturally replicate the cleaning power of the atmosphere makes them one of the most effective air purification technologies available today.
What Are Ions?
Ions are charged particles in the air that are formed in nature when enough energy acts upon a molecule such as carbon dioxide, oxygen, water or nitrogen to eject an electron from the molecule leaving a positively charged Ion. The displaced electron attaches itself to a nearby molecule, which then becomes a negatively charged Ion. It is the negative ion of oxygen that affects us the most. Remember that feeling you’ve experienced near a waterfall or high in the mountains? Those are two places that thousand of negative ions occur. They create an effect on human biochemistry. The normal Ion count in fresh country air is 2,000 to 4,000 negative Ions per cubic centimeter (about the size of a sugar cube). At Yosemite Falls, you’ll experience over 100,000 negative Ions per cubic centimeter; on the other hand, the level is far below 100 per cubic centimeter on the Los Angeles freeways during rush hour.
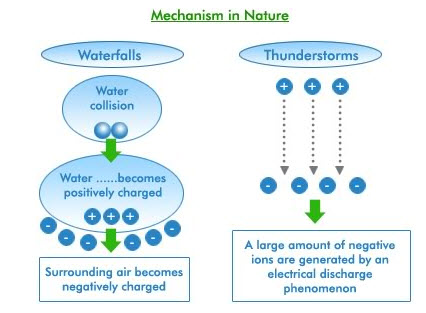
What Are The Benefits of Using Negative Ions?
The negative ions cause microscopic particles (particulates) floating in a room, that often cause people to have allergic reactions, to clump together and fall to the floor where they can be vacuumed up. This is due to an electrostatic charge between the negative ions and other air molecules and particles in the air. Negative ionizers have been used for years to help rid closed indoor environments of allergens such as dust particles, animal dander, pollen, mold spores, cigarette smoke, cigar smoke, particulate matter, etc. floating in the air. According to the book,The Ion Effect, negative ions are effective for allergies, asthma, catarrh, hay fever, sinusitis, eczema, burns, emphysema, and even as a substitute for tranquilizers.